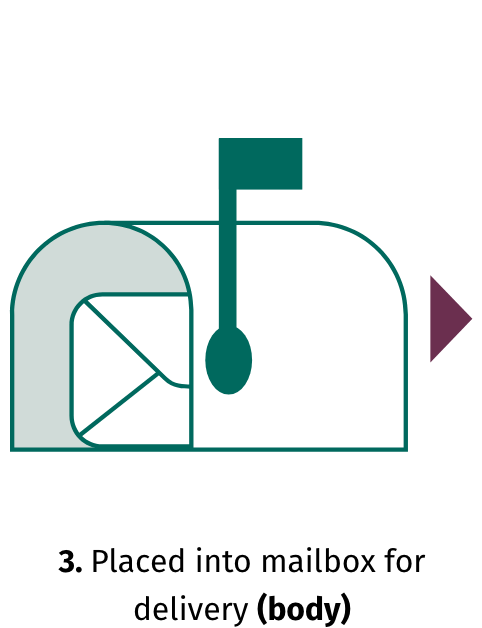
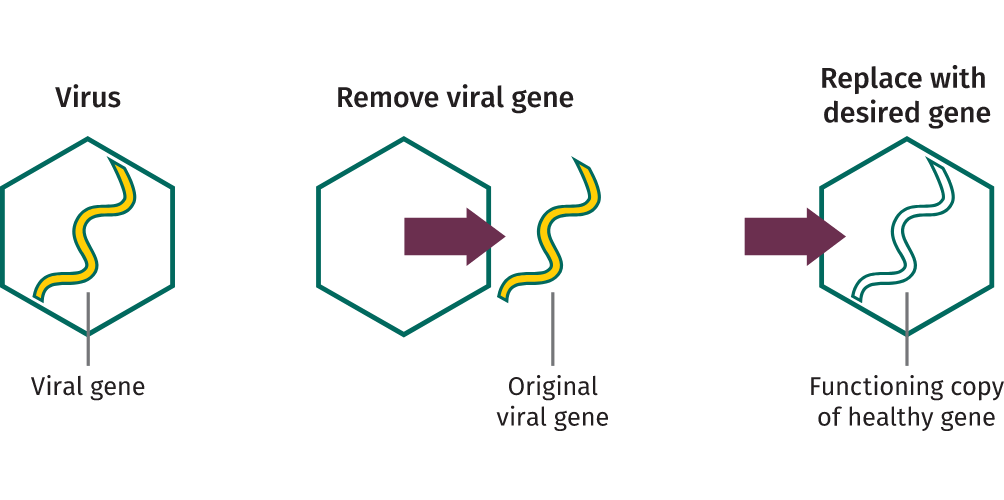
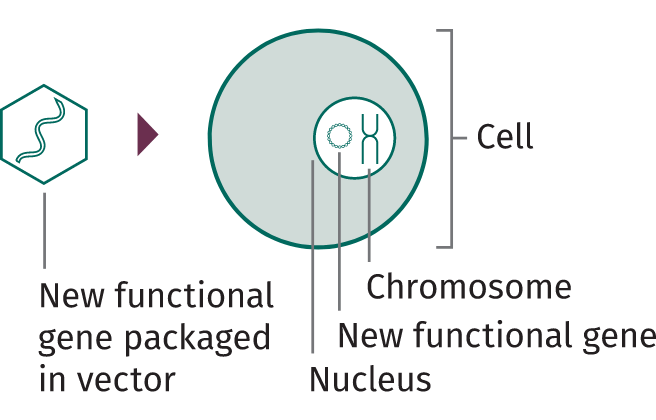
© 2024 Spark Therapeutics, Inc. N-HEM-US-700002-12
Spark, Spark Therapeutics and its Design, Hemophilia Forward, Hemophilia Forward and its Design are registered trademarks of Spark Therapeutics, Inc. in the United States.
Disclaimer: Nothing on HemophiliaForward.com should be construed as medical advice or replace medical advice, medical visits, or recommendations from a healthcare professional. Patients should always consult with a doctor or other healthcare professional for medical advice.